How Your Gut Microbiome Changes Your Drug Response and Side Effects
 Apr, 13 2026
Apr, 13 2026
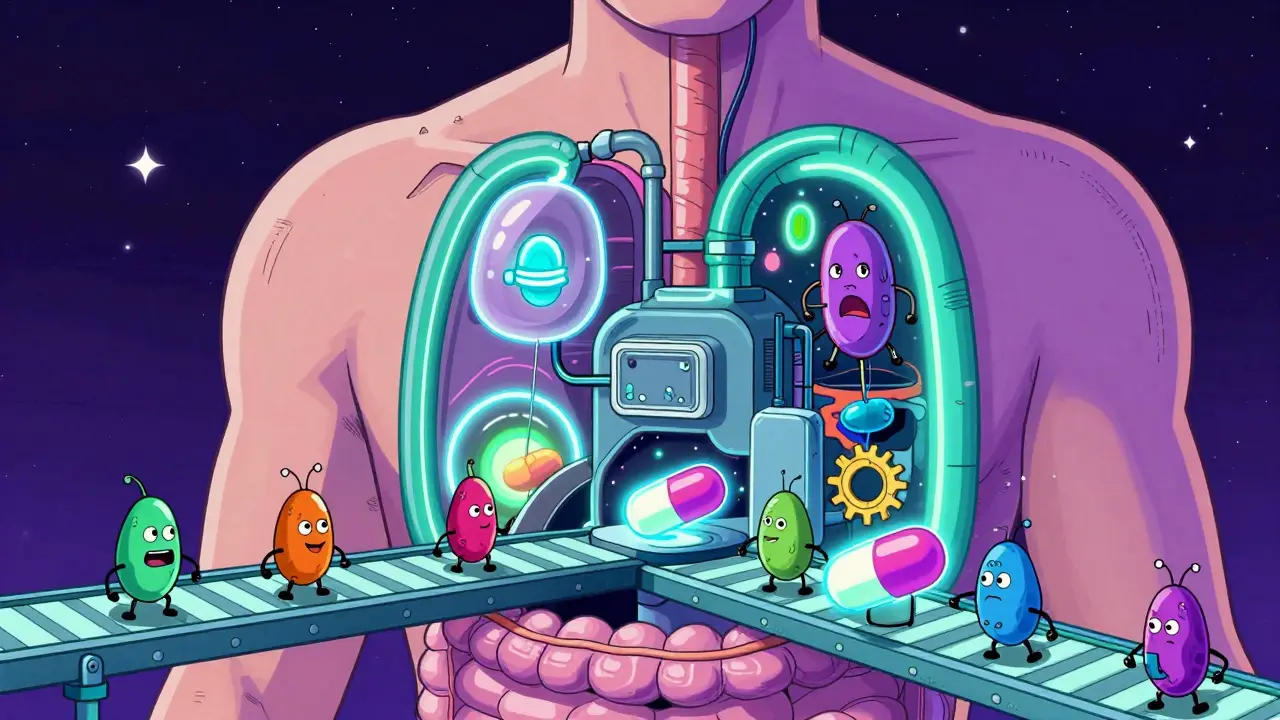
Gut-Drug Interaction Simulator
Select a medication to see how the gut microbiome alters its chemical structure and the resulting clinical outcome.
Irinotecan
Beta-glucuronidase reactivation
Severe intestinal damage / diarrhea
Mechanism Breakdown:
The body neutralizes Irinotecan into SN-38-glucuronide. However, specific gut bacteria produce beta-glucuronidase which "undoes" this neutralization, reverting it back into the toxic SN-38 form.
Ever wonder why a medication that works like a charm for your neighbor makes you feel sick? Or why some people experience severe side effects from a dose that others barely notice? For decades, doctors blamed genetics or liver function. But there is a hidden player in your gut that often holds the real answer. Your gut microbiome is the collective community of trillions of microorganisms living in your gastrointestinal tract. It doesn't just help you digest food; it acts like a second liver, chemically altering the drugs you swallow before they even hit your bloodstream.
This isn't just a theoretical idea. Research from Yale University found that gut bacteria can transform common medications into harmful compounds. In some cases, these microbes are responsible for 20% to 80% of the toxic metabolites circulating in a patient's body. If your specific bacterial makeup is different from someone else's, your body might produce a "toxic version" of a drug that another person's body simply ignores. This shift is turning the world of pharmacology on its head and paving the way for a more personalized approach to medicine.
Key Takeaways: The Gut-Drug Connection
- Bacteria in your gut can activate "prodrugs" or turn safe medications into toxic metabolites.
- Individual differences in microbiome composition explain why drug side effects vary so wildly between people.
- Specific enzymes, like bacterial beta-glucuronidase, are directly linked to severe side effects in chemotherapy patients.
- Antibiotics can accidentally change how other medicines work by wiping out the bacteria that metabolize them.
- Precision medicine is moving toward microbiome screening to predict and prevent adverse drug reactions.
How Bacteria Actually Change Your Meds
When you take a pill, it doesn't just vanish into your blood. It travels through the colon, where bacterial density is incredibly high-reaching up to 1011 to 1012 CFU/mL. Here, the microbiome uses various enzymatic tools to chop up or add pieces to drug molecules. This process is called drug metabolism, and in the gut, it happens through several specific chemical reactions.
Experts like Ian D. Wilson and Jeremy K. Nicholson have cataloged seven primary ways these bacteria manipulate drugs: acetylation, deacylation, decarboxylation, dehydroxylation, demethylation, dehalogenation, and conjugate hydrolysis. To put it simply, the bacteria are treating your medication like a food source or a chemical intruder, changing its structure in the process.
Some drugs are designed as prodrugs, meaning they are inactive when you swallow them and require bacteria to "turn them on." For example, the drug prontosil is virtually useless unless bacterial azoreductase enzymes break it down into sulfanilamide. If you've recently taken a heavy course of antibiotics that wiped out those specific bacteria, the drug's efficacy can plummet from 90% down to just 12%.
When the Microbiome Turns a Drug Toxic
The real danger arises when the microbiome converts a safe drug into a toxic one. A striking example is seen in oncology. Many cancer patients receive irinotecan, a chemotherapy drug. The body normally neutralizes this drug into a harmless form called SN-38-glucuronide. However, certain gut bacteria produce an enzyme called beta-glucuronidase that "undoes" this neutralization, turning the drug back into the toxic SN-38. This causes severe, dose-limiting diarrhea in 25% to 40% of patients.
This isn't limited to cancer meds. Researchers found that for one specific antiviral drug, gut microbes produced 73% of the circulating toxic metabolite. This explains why 15-20% of patients experience severe reactions while the rest are fine. Even heart medications like digoxin are affected; a bacterium called Eggerthella lenta can actually inactivate the drug, leading to a 30% difference in how well the medication works from person to person.
| Drug Name | Microbiome Action | Clinical Outcome |
|---|---|---|
| Irinotecan | Beta-glucuronidase reactivation | Severe intestinal damage / diarrhea |
| Prontosil | Azo reduction (activation) | Loss of efficacy if bacteria are absent |
| Digoxin | Reduction by E. lenta | 30% variability in drug effectiveness |
| Lovastatin | Metabolism dependency | 35% reduced efficacy after antibiotics |
| Clonazepam | General microbial metabolism | 40-60% higher plasma levels in germ-free models |

The Antibiotic Paradox
We usually think of antibiotics as a way to kill "bad" bacteria, but they are a sledgehammer that hits the "good" metabolic bacteria too. This creates a dangerous feedback loop. When you deplete your microbiome, you aren't just losing digestion help; you're changing your pharmacokinetics-the way your body absorbs, distributes, and eliminates drugs.
Take lovastatin, a common cholesterol medication. Research shows that patients on long-term antibiotic treatment may see a 35% drop in the drug's efficacy because the bacteria needed to help process the medication are gone. Similarly, in animal studies, removing gut bacteria reduced the birth defects caused by nitrazepam by 78%, proving that the "toxic" version of the drug was only created because of the microbiome.
Moving Toward Precision Medicine
So, how do we fix this? The goal is to move away from "one-size-fits-all" dosing. Instead of guessing why a patient is reacting poorly, doctors may soon screen your microbiome before prescribing. This is the core of precision medicine: matching the drug to the patient's internal ecosystem.
Currently, the industry is exploring a few paths:
- Metagenomic Sequencing: Testing a fecal sample (costing roughly $300-$500) to identify which metabolic genes you have.
- Enzyme Inhibitors: Creating "blocker" drugs. For example, beta-glucuronidase inhibitors are in Phase II trials and have already shown a 60% reduction in chemotherapy-induced diarrhea.
- Custom Probiotics: Designing specific bacterial strains to help patients metabolize drugs more efficiently.
Large pharmaceutical companies like Pfizer and Merck are already adding microbiome screening to early-stage clinical trials. While it adds a few million dollars to development costs, it's a drop in the bucket compared to the hundreds of millions lost in lawsuits or recalls when a drug causes unexpected adverse events in a large portion of the population.

The Future of Personalized Dosing
Within the next few years, we might see dosing algorithms that factor in your bacterial profile. Imagine a pharmacist saying, "Based on your gut flora, we need to lower your dose of this medication by 20% to avoid toxicity," or "You need a specific probiotic for two weeks before starting this drug to make it work."
The NIH has already put $14.7 million into the Gut Microbiome and Pharmacology Initiative to turn this research into real-world clinic tools. While we aren't there yet, the evidence is clear: the bacteria in your gut are just as important as the genes in your DNA when it comes to how you respond to medicine.
Can probiotics help reduce drug side effects?
Potentially, yes. Researchers are currently developing personalized probiotic formulations designed to modulate specific drug metabolism pathways. While general over-the-counter probiotics may not target specific drug-metabolizing enzymes, clinical trials are underway to create targeted strains that can either activate prodrugs or prevent the formation of toxic metabolites.
Do antibiotics always interfere with my other medications?
Not always, but they can. Antibiotics change the composition of your gut microbiome. If your medication relies on bacterial enzymes for activation (like some prodrugs) or if the microbiome helps clear a drug from your system, antibiotics can either make the medication less effective or increase its toxicity in your bloodstream.
Which drugs are most affected by the microbiome?
Oncology drugs (like irinotecan), certain antivirals, and heart medications (like digoxin) are heavily influenced. According to a 2023 Nature analysis, there are at least 117 drugs where gut bacteria significantly alter pharmacokinetics, with the majority showing reduced efficacy in some patients.
Is there a test I can take to see my microbiome's effect on drugs?
Currently, metagenomic sequencing is the most accurate way to identify metabolic genes in your gut, but this is mostly used in research and high-end clinical settings rather than standard pharmacies. However, microbiome screening is becoming more common in pharmaceutical clinical trials.
Why does the microbiome change drug metabolism more in the colon than the small intestine?
Bacterial density is significantly higher in the colon, reaching 1011 to 1012 CFU/mL. Additionally, the transit time in the colon is much slower (typically 24-72 hours), giving the bacteria more time to interact with and chemically transform the pharmaceutical compounds.
Next Steps and Troubleshooting
For Patients: If you feel your medication isn't working or you're experiencing unusual side effects, don't stop taking your meds. Instead, ask your doctor if your recent use of antibiotics or a change in diet could be affecting the drug's efficacy. Mention the potential for microbiome-drug interactions if you are on high-risk medications like chemotherapy.
For Healthcare Providers: Start considering the "microbial history" of your patients. A patient who has undergone frequent broad-spectrum antibiotic therapy may have a depleted microbiome, potentially altering the response to statins or the activation of certain prodrugs. Stay updated on the emerging FDA and EMA guidelines regarding microbiome interaction studies for drugs with narrow therapeutic indices.
Clare Elizabeth
April 15, 2026 AT 09:29this is so cool!! imagine a world where we actually get the right dose the first time around without all the guessing games 🚀 just love seeing science move toward making things a bit more human and personalized for everyone
Sam Dyer
April 16, 2026 AT 06:38Typical ivory tower stuff. They're finally "discovering" that one size doesn't fit all? No kidding! 🙄 My old man took the same blood pressure pill as his buddy and one felt great while the other felt like a zombie. It's common sense, not rocket science, but sure, let's spend millions of tax dollars to tell us our gut bacteria are different. Give me a break! 🇺🇸
mimi clouet
April 17, 2026 AT 02:21Actually, it's not just about the bacteria themselves but the specific metabolic pathways they trigger! 🤓 I've read that the beta-glucuronidase enzyme is just the tip of the iceberg when it comes to how our microbiome interacts with pharmacokinetics. It's honestly fascinating how a simple change in diet can shift these levels 🌟✨
Catherine Mailum
April 18, 2026 AT 12:11oh wow because clearly the medical industry is just *so* famous for its efficiency and not making us guinea pigs for thirty years first 🙄 just great that we have to pay $500 for a poop test now to make sure our meds don't kill us
S.A. Reid
April 18, 2026 AT 22:17One must wonder if this "precision medicine" is merely a sophisticated veil for further corporate surveillance of our biological data. It is quite plausible that by sequencing our microbiomes, these pharmaceutical conglomerates are simply cataloging our vulnerabilities to better manipulate the market. I find it rather quaint that some believe this is purely for patient benefit. The intersection of biotechnology and data harvesting is a precarious slope indeed.
Jasmin Stowers
April 20, 2026 AT 18:47makes a lot of sense actually. always wondered why some meds just didn't hit the same for me
Randy Ryder
April 21, 2026 AT 10:07The implications for the first-pass metabolism are staggering. If we can effectively modulate the enterohepatic circulation via targeted microbial interventions, we could theoretically optimize the bioavailability of narrow-therapeutic-index drugs. I'm particularly interested in whether the metabolic flux can be shifted using non-absorbable polymers to shield the drug from bacterial enzymatic degradation in the colon.
Kenzie Evans
April 22, 2026 AT 20:16Who cares about the science when the actual implementation is probably going to be a disaster? Only rich people will afford these "custom probiotics" while the rest of us keep getting diarrhea from chemo. This whole post is just fluff to make Big Pharma look like they care about us. Utterly useless.
Ikram Khan
April 23, 2026 AT 12:35Wait, so my antibiotics could actually make my other meds stop working?! That is absolutely wild! 😱 I had no idea my gut was basically a second pharmacy in there. This is such a game changer for how I think about my health! 💊✨
Anurag Moitra
April 24, 2026 AT 13:48It is imperative that we encourage patients to maintain a detailed record of their antibiotic history. This information is vital for clinicians to understand potential drug-microbiome interactions and to avoid suboptimal therapeutic outcomes for the patient
Brooke Mowat
April 25, 2026 AT 04:30It's just so poetic really... like we're these walkin' ecosystems and we aren't even the boss of our own bodies lol. Just a bunch of tiny bugs decidinnn if the medicine works or not. Honestly it's a vibe to realize we're just a vessel for trillions of little friends doin' their own thing in the dark 🌿✨
Mark Dueben
April 26, 2026 AT 06:31Maybe we could all try to be a bit more mindful of how we talk about these "toxic metabolites." It's a bit scary for some people, so framing it as an opportunity for better care might be more inclusive for those who are currently struggling with their treatments.
rupa das
April 27, 2026 AT 04:37precision medicine is just a fancy word for overpriced tests that wont actually change anything in a real clinic
Milo Tolley
April 27, 2026 AT 18:26The sheer pharmacokinetic volatility described here is simply... UNACCEPTABLE!!! How have we been ignoring the role of the gut microbiota in the titration of these compounds for so long??? It is a categorical failure of the medical establishment to ignore the metabolic synergy between the host and the commensal flora!!! This is absolute madness!!!
Becca Suttmiller
April 28, 2026 AT 06:19I appreciate the detailed breakdown of the different enzymes. It helps to understand the biology without feeling overwhelmed by the medical jargon.